Base saturation is something more often than not, too quickly overlooked on a soil test. Base saturation shows the percentage quantity of calcium, magnesium, potassium, and hydrogen in the soil. The lower the saturation gets for calcium and magnesium, the more nitrogen will build up in the soil. This is what causes pH to become acidic. That hydrogen is blocking the movement of nutrients in the soil and to the plant that in turn hinders growth and causes the fertilizer to become bound up and go to waste.
Recommended liming and fertilizer practices will vary for soils with widely differing cation exchange capacities. For instance, soils having a high CEC and high buffer capacity change pH much more slowly under normal management than low CEC soils. Therefore, high CEC soils generally do not need to be limed as frequently as low CEC soils; but when they do become acidic and require liming, higher lime rates are needed to reach optimum pH.
CEC can also influence when and how often nitrogen and potassium fertilizers can be applied. On low CEC soils (less than 5 meg/20000g), for example, some leaching of cations can occur. Fall applications of ammonium N and potassium on these soils could result in some leaching below the root zone, particularly in the case of sandy soils with low CEC subsoils. Thus, spring fertilizer application may mean improved production efficiency. Also, multi-year potash applications are not recommended on low CEC soils. Higher CEC soils (greater than 10 meg/100g), on the other hand, experience little cation leaching, thus making fall application of N and K a realistic alternative. Applying potassium for two crops can also be done effectively on these soils. Thus, other factors such as drainage will have a great effect on the fertility management practices used on high CEC soils (Purdue University Extension).
Base Saturation:
- Exchangeable cations can be divided into two groups:
- Bases
- Acids
- Every CEC binding site must have a cation bound to it to maintain electroneutrality
- The soil pH will be affected by whichever cations predominate on these exchange sites
- More base cations create more alkaline soil
- More acid cations create more acidic soil
Calcium & Base Saturation
Because calcium has an atomic weight of 40 units and hydrogen only has an atomic weight of 1, it is very easy for calcium to displace the hydrogen molecule from the CEC binding site and take its place. This negates the need for a carbon molecule as is used in limestone to strip hydrogen from the soil.
The optimal levels for base saturation are 65-75% calcium, 15% magnesium, 2-4% potassium, and about 10-15% hydrogen. At these levels, soil pH is balanced right around 7.0 neutral state. But, most importantly, the nutrients in the soil such as nitrogen, phosphorus, and all the key elements of plant growth. When the base saturation is balanced the cation exchange in the soil is at its most efficient. Additionally, when these levels are balanced, soil is more porous, meaning there is less compaction, better water disbursement, and retainment of that water, which leads to a healthier, stronger crop. Essentially the base saturation of calcium and magnesium in the soil is directly correlated to the soil pH, hence the reason for using calcitic lime or dolomitic lime.
How Liquid Calcium is Applied
Soil pH is a measure of the acidity in the soil. That acidity is determined by the amount of the hydrogen (H+) built up in the soil. Hydrogen is an acidic cation whereas calcium and magnesium are basic cations. Balancing soil pH is simply balancing the levels of the cations at a state that maximizes nutrient availability in the soil. Soile is the nutrient storehouse for plants. We measure the cation amounts in the soil based on a 100% soil capacity, such as discussed in the base saturation section. The higher the percentage of hydrogen in the soil compared to other primary cations, calcium, and magnesium, the lower the soil pH becomes. However, when calcium or magnesium are added to the soil, it raises each of their respective percentage levels, in turn lowering hydrogen levels and raising soil pH.
Example:
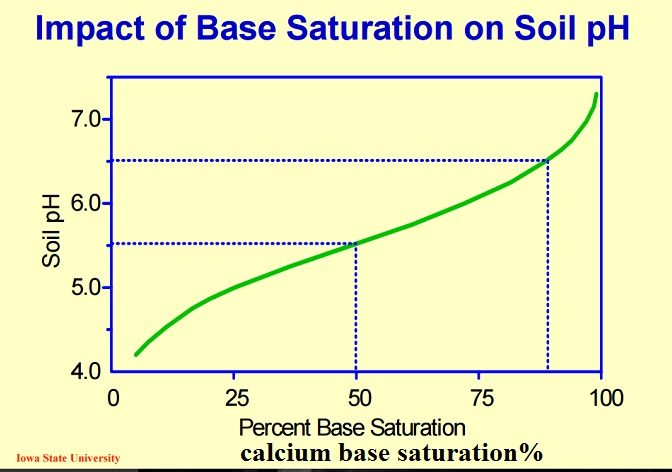
Soil pH: 5.5
Base saturation levels: Ca++: 50% Mg++: 12% K+: 3% H+: 35%
If calcium is added to the soil to raise the calcium base saturation by 25% to the necessary level, the following result will occur:
Soil pH 6.8
Base saturation levels: Ca+: 75% Mg+: 12% K+: 3% H+: 10%
Notice the difference being the additional 25% calcium introduced to the soil lowered the overall percentage of hydrogen by the same quantity, In turn balancing pH at a neutral state or near neutral state.
Effects of Liquid Calcium
Liquid calcium works by quickly and effectively adding the needed calcium to the soil, raising base saturation and balancing pH. This is what is happening when a calcitic lime is added to the soil. The difference being the limestone must take 6 months – 1 year to breakdown. During this time, the stone is releasing soluble calcium into the soil and raising pH. However, only a small percentage of limestone actually becomes available calcium, with the majority settling out in the soil causing hard plates and soil compaction over time.
Liquid calcium eliminates those issues by bypassing the breakdown process and eliminating harmful sediment that can hurt soil conditions in the long term. To put this simply, limestone and liquid calcium in the end administer the same results, which is adding calcium to the soil to raise pH, increase cation exchange in the soil, rid the soil of sage weeds, and boost overall plant growth and health. Liquid calcium is simply the more effective way of attaining those results. The reason this method seems to be new to the industry, although it is not, is because for years, it has not been cost-effective to do so. However, with current day ag prices plut the cost of trucking and spreading, liquid calcium is not only the better option for correcting soil pH, but also the more affordable option.


